Our lab is focused on advanced artificial intelligence (AI) and quantitative imaging technology to advance disease diagnosis, prognosis, and treatment evaluation. We aim to develop and deploy our algorithms in healthcare workflow. Our current research includes self-supervised AI algorithm development for ophthalmic diagnosis, medical federated learning, multi-modal AI models, and quantitative optical coherence tomography (OCT) and OCT angiography (OCTA) imaging biomarker development.
Our current research is funded by NIH-NEI R21 and R15 mechanism, Wake Forest Center for AI Reserach (CAIR) – UNC Charlotte AI4Health Center seed funding, and UNC Charlotte Faculty Research Grant.
ONGOING RESEARCH PROJECTS:
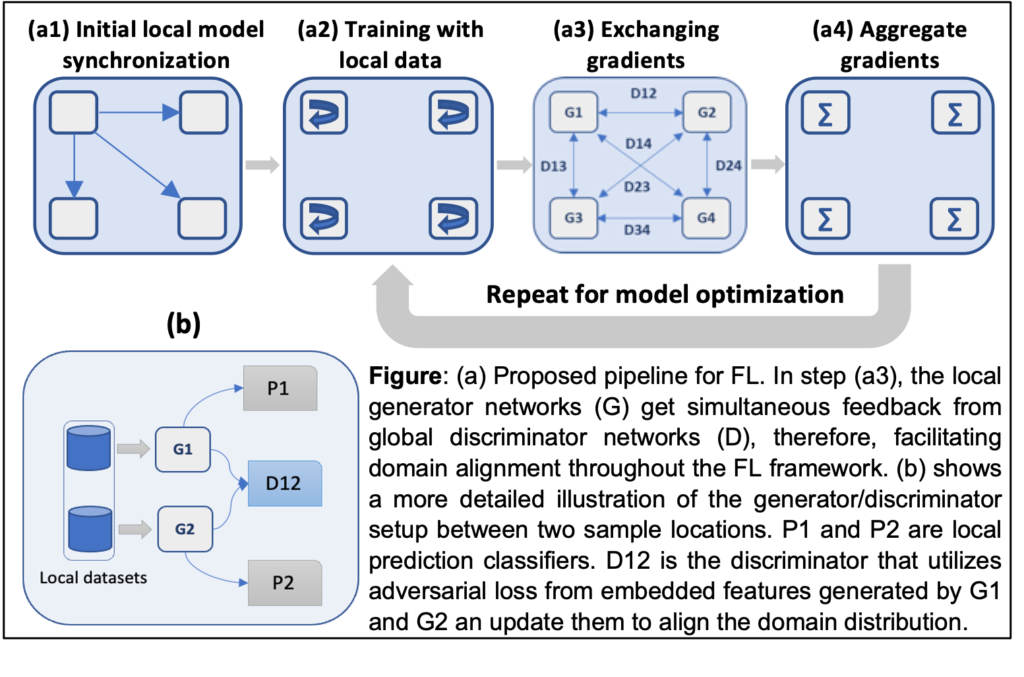
Distributed approaches for training medical ML models:
- This project develops novel domain adaptive federated learning models to train medical ML models.
- Fundamental research on distributed model training, granular privacy, and security-performance tradeoff.
- Fundamental exploration on how to address challenges in federated learning such as noisy labels, heterogeneous distribution from a wide-spectrum of sub-population data.
Multi-modal (image and language data) foundational models in ophthalmic diagnosis
- This project aims to design self-supervised, attention-driven, end-to-end learning models to extract comprehensive features from extensive, unlabeled, multi-modal datasets (image and text).
- Fundamental research on distributed model training, granular privacy and security-performance tradeoff.
- Vision-language embeddings – fundamental research on how to learn representations together.
- Innovation within federated learning by proposing privacy-preserving, domain-adaptive algorithms combined with novel domain alignment methods to effectively address and reconcile domain shifts resulting from the varied data (multi-modal image and text) distributions across multiple institutions.
- Impacts the clinical management of ophthalmic diseases and provides textual insights to AI models.
Functional retinal biomarkers for neuro-retinal degenerative disease
- This project establishes fundamental understanding of neurovascular coupling in human retina through stimulus-evoked neural and hemodynamic intrinsic optical signals (IOS), associated with retinal photoreceptor responses.
- This project develops a novel generative AI-based optical coherence tomography (OCT) angiography (OCTA) construction algorithm utilizing spatial vascular connectivity and heuristic contextual guidance to enable functional imaging of stimulus evoked hemodynamic IOS in single scan volumetric OCT data from commercial clinical devices.
NEI R15 – Distributed approaches to train machine learning models in diabetic retinopathy
This project aims to establish distributed federated learning (FL) approaches for training robust, clinically deployable machine learning (ML) models for, i) multi-class classification of DR, and ii) prediction of proliferative DR (PDR) progression, in optical coherence tomography (OCT) angiography (OCTA). DR is one of the leading causes of severe vision loss.
Early detection, prompt intervention, and reliable assessment of treatment outcomes are essential to prevent irreversible vision loss from DR. Quantitative OCTA analysis and OCTA-ML models have recently been applied to diagnose, classify, and understand the progression trends of DR. Despite promising results, the clinical utility of OCTA based diagnostic algorithms is not yet fully determined, due to small OCTA data-cohorts in clinical institutions, and the lack of wide-spread validation. More specifically, a major limitation of OCTA-ML models is the need for large amounts of well curated datasets from a diverse sub-population for robust performance. Moreover, efforts towards large, centralized datasets for ML research are hindered by significant barriers to data sharing and privacy concerns.
In this project, we aim to establish novel federated ML approaches, where the model training is distributed across institutions instead of sharing patient data and only the model parameters are shared with a central server. This enables gaining insights collaboratively, e.g., in the form of a consensus model, without moving patient data beyond the firewalls of the institutions. Three data cohorts from the Stanford University, University of Illinois Chicago (UIC), and National Taiwan University (NTU) will be used to test the hypothesis that the accuracy of the OCTA-ML models using federated approach is more robust than models built on single institutional datasets.
Success of this project will establish distributed ML model training approaches and pave the way towards using quantitative OCTA features for early DR detection, objective prediction and assessment of treatment outcomes.